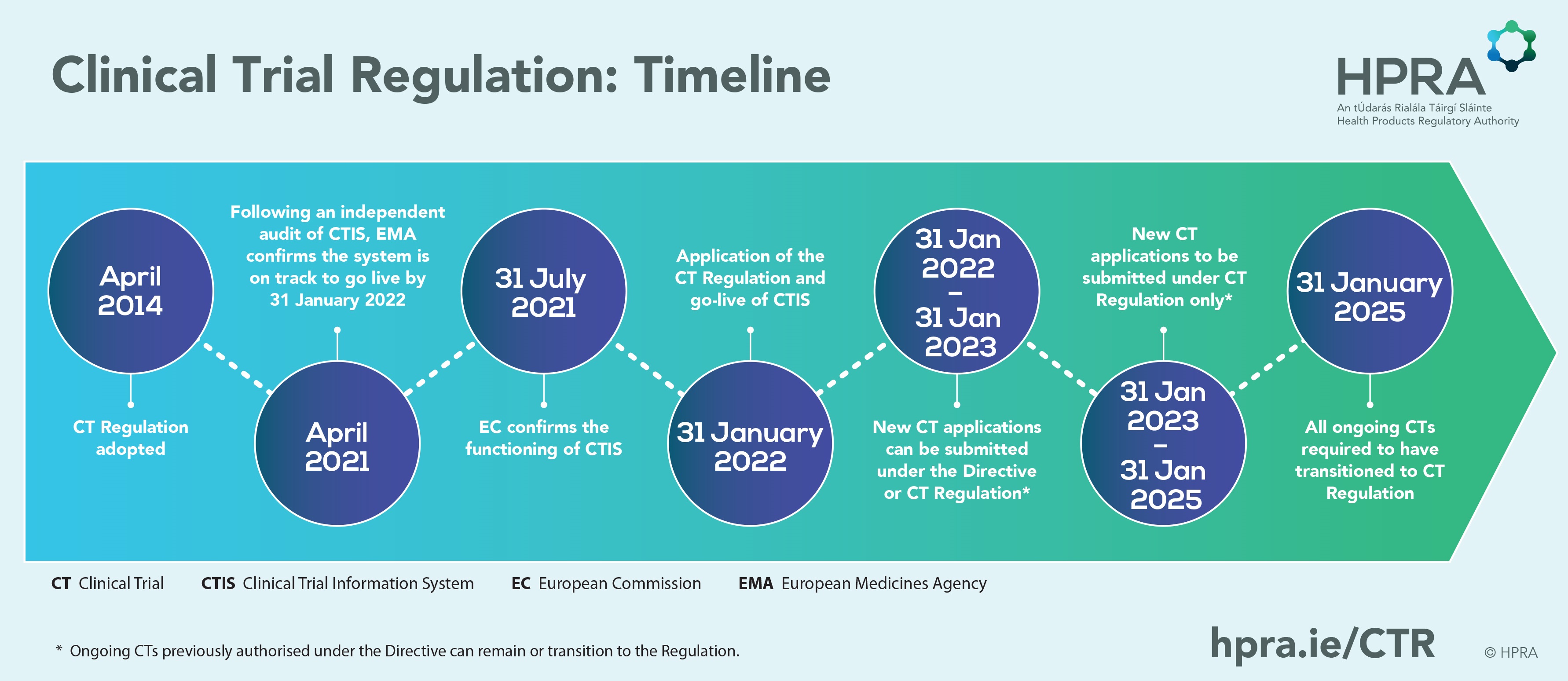
EMA & FDA Approvals and Recommendations in 2020 for Oncology Drugs and Diagnostics/Devices | CATO SMS
Regulatory Affairs 101: Introduction to Investigational New Drug Applications and Clinical Trial Applications
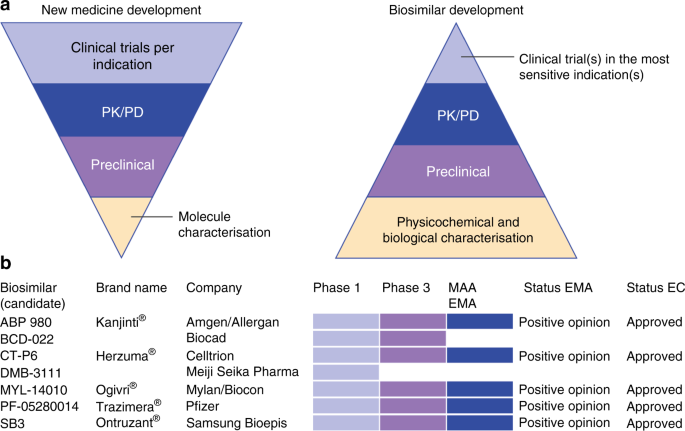
The arrival of biosimilar monoclonal antibodies in oncology: clinical studies for trastuzumab biosimilars | British Journal of Cancer

When innovation outpaces regulations: The legal challenges for direct‐to‐patient supply of investigational medicinal products - Malone - 2022 - British Journal of Clinical Pharmacology - Wiley Online Library

Current landscape of clinical development and approval of advanced therapies: Molecular Therapy - Methods & Clinical Development
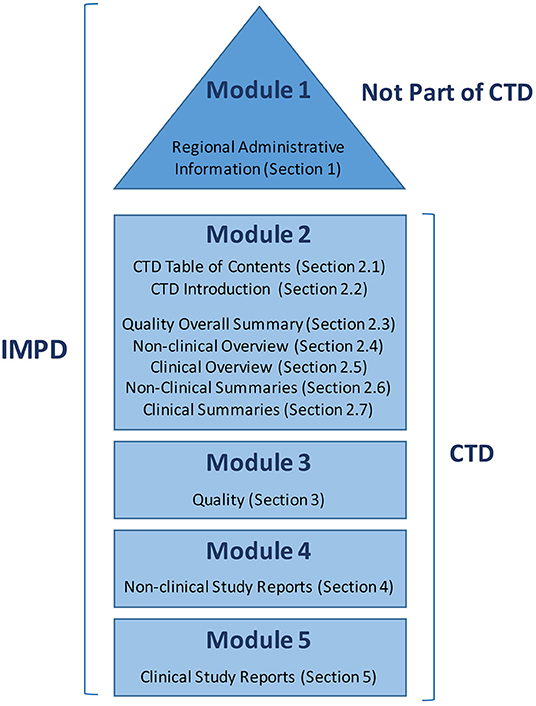
Frontiers | Transitioning From Preclinical Evidence to Advanced Therapy Medicinal Product: A Spanish Experience
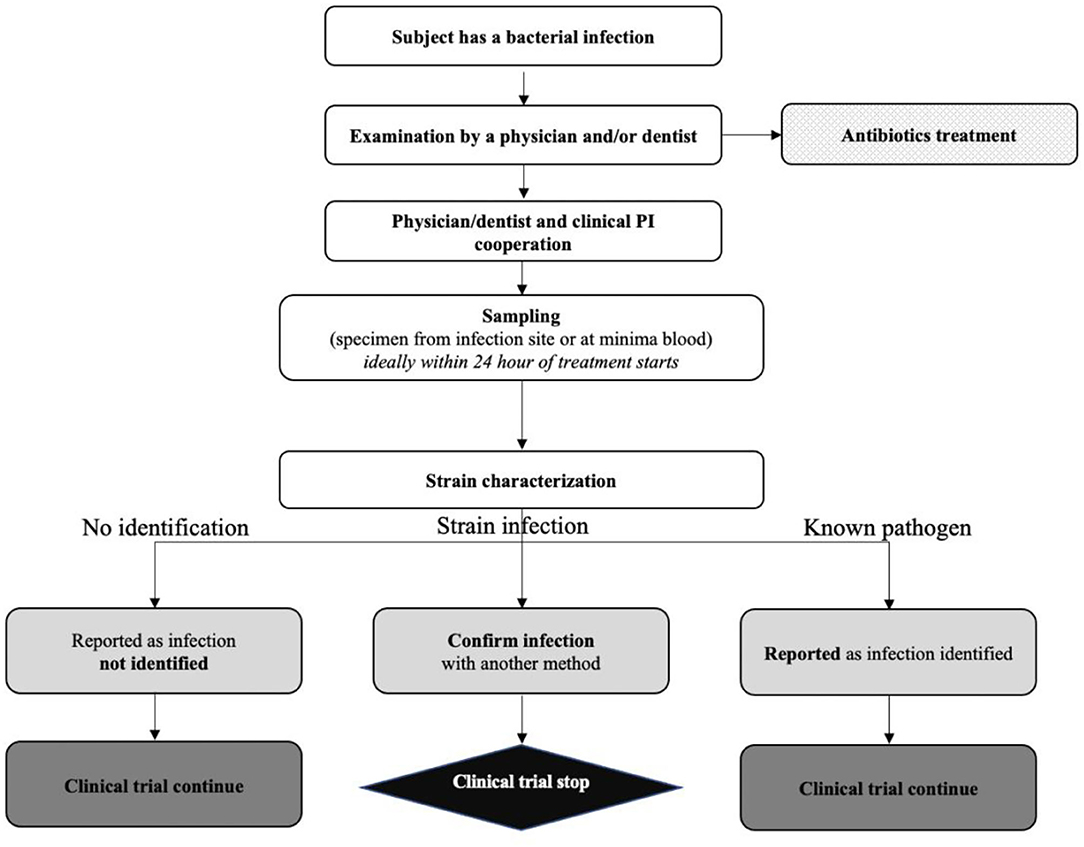
Frontiers | Entering First-in-Human Clinical Study With a Single-Strain Live Biotherapeutic Product: Input and Feedback Gained From the EMA and the FDA

Tag | CRO, Clinical, TMF, eTMF, trial master file, inspection, audit, audit ready, inspection ready, Clinical Research Organisation, regulation, EU, EMA, MHRA, Andy FIsher | Pharma IQ

Comparison of regulatory pathways for the approval of advanced therapies in the European Union and the United States - Cytotherapy